European Journal of Neurodegenerative Diseases 2017; 6(1) January-June: 1-5
IN THE BRAIN, VARIOUS INFLAMMATORY MEDIATORS CAN BE RELEASED THROUGH THE ACTIVATION OF MAST CELLS
Y. Gu 1 and D.K. Yang 2
1 Radiotherapy Ward 3, The First Affiliated Hospital of Zhengzhou, Zhengzhou, Henan, China;
2 Compositive Thermotherapy Center, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan, China.
*Correspondence to:
Dr. D.K. Yang,
Compositive Thermotherapy Center,
The First Affiliated Hospital of Zhengzhou University,
Zhengzhou, Henan,
450052 China.
ABSTRACT
In humans, mast cells (MCs) are ubiquitous in tissues and express numerous surface receptors that allow them to respond to various stimuli and participate in many diseases, including allergies, inflammation, infections, and autoimmune and brain diseases. The activation of MCs in the brain can induce neuroinflammation with release of proinflammatory cytokines and chemokines and activates other compounds such as histamine and serotonin (5-HT), which play an important role in the central nervous system (CNS). The immediate degranulation of MCs leads to the production of histamine and 5-HT which contributes to the inflammatory process. Activated MCs release numerous neuroinflammatory molecules such as the cytokines tumor necrosis factor (TNF), IL-1, IL-6, vascular endothelial growth factor (VEGF), transforming growth factor beta (TGFb), and CXCL8, amongst others. The inflammatory effect of MCs can overlap with that of peripheral circulating monocytes, amplifying neuroinflammation and neurodegeneration.
KEYWORDS: mast cell, neuroinflammation, brain, neurodegeneration, cytokine, chemokine
INTRODUCTION
The immune system plays an important role in brain diseases. Immune cells are ubiquitous and therefore also reside in the meninges where they act as guardians to protect the body against microorganisms (1). In trying to defend the body, immune cells produce inflammatory molecules such as cytokines, metalloproteinases, arachidonic acid products, and other compounds, that mediate brain pathology (2). Mast cells (MCs) are immune cells that mediate innate immunity and acquired immunity. They are proinflammatory effector cells, that reside in high numbers within the central nervous system (CNS) and can play critical roles in the development of inflammation in many disorders (3). MCs are found throughout the body, including the brain. When they are activated in the brain, MCs release biological mediators such as cytokines/chemokines, leukotrienes, prostaglandins, and stored enzymes, all which play an important role in brain disorders (3,4).
Neuroinflammation is associated with neurological and cognitive effects with mechanisms that are still unclear. MCs can mediate CNS immune responses and play a role in neuroinflammation and in several neurological diseases such as Alzheimer’s disease (AD) (5), multiple sclerosis (6), and Parkinson’s disease (7).
DISCUSSION
MCs are immune cells of bone marrow origin that are involved in the pathophysiology of many allergic and non-allergic diseases. Moreover, they participate in many pathological processes, including brain diseases and autoimmune diseases. MCs mediate brain damage in stress and neuropsychiatric disorders by releasing inflammatory mediators which can be of two types: those secreted immediately, such as proteases, and those secreted after a few hours, such as cytokines and chemokines (8).
MCs contribute to almost all aspects of neuroinflammation by releasing inflammatory mediators (9). The cytokines released by MCs that are the most reported to induce neuroinflammation are tumor necrosis factor (TNF), IL-1, IL-6, vascular endothelial growth factor (VEGF), and transforming growth factor beta (TGFb) (10). The chemokines CXCL8 and CCL2, 3, and 4 also participate in neuroinflammation (11). Some cytokines, such as IL-1, can have an autocrine effect by activating microglia through toll like receptor (TLR) 4 which amplifies neuroinflammation (12). Pro-inflammatory cytokines induce capillary permeability and leak, and vascular dilation in brain tissue, and activate several inflammatory genes. In diseases such as AD, MCs react to plaque formations by producing various cytokines and chemokines, facilitating the permeability of the blood-brain barrier (BBB) (13).
TNF is the only cytokine stored in the granules of MCs which can be released by degranulation immediately upon activation. In addition, TNF can be produced and secreted after several hours of protein synthesis. MC-derived TNF potently mediates the inflammatory reaction and sensitization of meningeal nociceptive receptors (14).
Microglia are immune cells that are similar to macrophages and are located in the CNS. Microglial cells are phagocytic immune cells that release a series of pro-inflammatory molecules, such as IL-1, TNF, and IL-6, when they are activated from endogenous or exogenous stimulus (15). Neuroinflammation and neurodegeneration can be amplified when the inflammatory effect of microglia overlaps with that of peripheral circulating monocytes.
MCs expose the TLR on their cell surface, which is capable of binding microorganisms with consequent cellular activation and an inflammatory response. Activated MCs immediately release (in seconds) preformed mediators such as histamine, protease, and TNF, and later (after hours) they secrete inflammatory cytokines and chemokines by protein synthesis (8) (Fig.1). Furthermore, MCs can also release arachidonic acid compounds, such as prostaglandin D2 (PGD2) and the leukotrienes C4, D4, and E4, which induce the slow reaction of anaphylaxis (SRS-A) (16). The microbial activation of the TLR on MCs leads to a series of cascading reactions, with the formation of NF-kB and the synthesis of pro-inflammatory cytokines such as IL-1 (17). MCs produce numerous vasoactive mediators and other molecules that induce inflammation. Many of these molecules can disrupt the BBB by promoting the passage of toxic molecules into the CNS (9). The release of inflammatory mediators from MC granules, such as histamine and serotonin (5-HT) (in rodents), occurs immediately after activation (18).
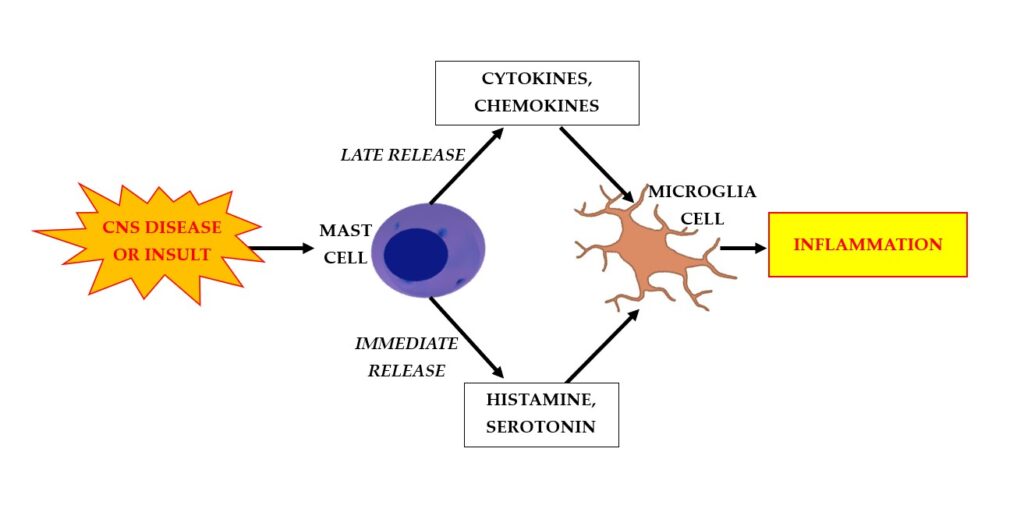
Fig. 1. Disease or insult in the central nervous system (CNS) activates mast cells (MCs) to rapidly release (after seconds) preformed mediators such as histamine and serotonin (5-HT), and later (after hours), to secrete inflammatory cytokines and chemokines. This process results in a cascade of further reactions that activates microglia cells and results in inflammation.
Histamine
Histamine is a biogenic amine that was first discovered by Windaus and Vogt in 1907 and is linked to the anaphylactic reaction. It is responsible for energy homeostasis, smooth muscle constriction, vasodilation, immunoregulation, and other functions (19). Histamine is ubiquitous in tissues and is contained in basophils, dendritic cells, T cells, platelets, neurons, and glial cells, and has important effects on the immune response and blood cells (20).
In MCs, histamine is rapidly synthesized and stored in specific granules from which it is subsequently released. Histamine is synthesized more slowly by immune cells such as T cells and is synthesized from the amino acid L-histidine via the carboxylation reaction (21). It is inactivated by histamine-N-methyltransferase or diamine oxidase, which is not found in the brain.
In the CNS, histamine is produced only in histaminergic neurons, has a half-life of approximately 25 minutes, and circulates in the body as a hormone that mediates diverse functions including vascular activity, cellular maintenance, metabolism, and inflammation (22). At the microglial level, histamine can be pro- or anti-inflammatory, depending on the homeostatic state. Histamine acts through four G-protein-coupled receptors: H1R, H2R, H3R, and H4R that are found in immune cells (23).
In the brain, H1R and H2R receptors are located on neurons and glia, while the H3R receptor is exposed only by neurons. H1R and H2R are found in numerous quantities in inflammatory cells such as MCs. The H2R mediates the inhibition of basophil degranulation, the innate immune response, and the generation of cytotoxic T lymphocytes (19). In addition, the stimulation of H2R in neutrophils and eosinophils inhibits the release of lysosomal enzymes and modulates the migration of these cells (24). The cytokines IL-1, IL-3, IL-8, and GM-CSF activate MCs to produce low quantities of histamine (25). After stimulation of H2R in lymphocytes, the release of histamine causes a lack of maturation and antibody production in B cells and plasma cells. The stimulation of H2R on T cells leads to the production of inflammatory cytokines.
Through the activation of the H1 receptor, histamine is responsible for 50% of prostaglandin generation. PGD2 is a potent vasoconstrictor which is completely released 5 minutes after MC activation of anti-IgE (26). Antihistamines, such as the anti-H2R, act as analgesics, likely by blocking 5-HT through Ca2+ channels (27).
Serotonin (5-HT)
5-HT is a neurotransmitter involved in many pathophysiological functions of the organism. It is contained in the granules of rodent MCs, but not in those of humans. 5-HT is involved in the gut-brain axis, a two-way interaction that leads to inflammation and other symptoms.
5-HT is related to the cellular immune system; In fact, drugs used to treat the effects of 5-HT also affect the immune system. Activated immune cells in the brain can mediate neuroinflammation and psychiatric diseases and 5-HT modulates the immune system through the regulation of immune cells, such as T cells, macrophages, dendritic cells, platelets, and MCs (28). The noradrenergic system is mediated by norepinephrine, which controls brain functions related to negative emotions. Norepinephrine can modulate the function of the peripheral immune system through 5-HT (29). In fact, the 5-HT 5-HT1A receptor promotes the proliferation of T lymphocytes, and blocking this receptor leads to an inhibition of cellular immunity and the synthesis of cytokines produced by T cells (30).
5-HT is found in many cells, including neurons, and the reduced expression of this neurotransmitter in the brain can lead to depression and other pathologies. 5-HT inhibitors can reduce the proliferation of CD4+ T lymphocytes and increase CD8+ suppressor cells, resulting in immunosuppression (28). These methods are useful in transplant rejection and autoimmunity. Furthermore, 5-HT is involved in the degranulation and migration of MCs, resulting in the secretion of pro-inflammatory cytokines. Thus, 5-HT mediates the immune response and inflammation, as well as CNS functions, mood, depression, food intake, anxiety, learning disorders, and other behavioral characteristics.
CONCLUSIONS
The activation of brain MCs can induce neuroinflammation with the release of proinflammatory cytokines and chemokines and can also activate chemical mediators such as histamine and 5-HT which play an important role in the CNS. However, these concepts reported here require further studies to clarify the mechanisms of action for the reactions involved. The inhibition of MC activation could represent an important therapeutic strategy for several neuroinflammatory diseases.
Conflict of interest
The authors declare that they have no conflict of interest.
REFERENCES
- Filiano AJ, Gadani SP, Kipnis J. Interactions of innate and adaptive immunity in brain development and function. Brain Research. 2015;1617:18-27. doi:https://doi.org/10.1016/j.brainres.2014.07.050
- Farooqui AA, Horrocks LA, Farooqui T. Modulation of inflammation in brain: a matter of fat. Journal of Neurochemistry. 2006;101(3):577-599. doi:https://doi.org/10.1111/j.1471-4159.2006.04371.x
- Dong H, Zhang X, Qian Y. Mast Cells and Neuroinflammation. Medical Science Monitor Basic Research. 2014;20:200-206. doi:https://doi.org/10.12659/msmbr.893093
- Silver R, Silverman AJ, Vitković L, Lederhendler II. Mast cells in the brain: evidence and functional significance. Trends in Neurosciences. 1996;19(1):25-31. doi:https://doi.org/10.1016/0166-2236(96)81863-7
- Shaik-Dasthagirisaheb YB, Conti P. The Role of Mast Cells in Alzheimer’s Disease. Advances in clinical and experimental medicine: official organ Wroclaw Medical University. 2016;25(4):781-787. doi:https://doi.org/10.17219/acem/61914
- Kritas SK, Saggini A, Cerulli G, et al. Impact of Mast Cells on Multiple Sclerosis: Inhibitory Effect of Natalizumab. International Journal of Immunopathology and Pharmacology. 2014;27(3):331-335. doi:https://doi.org/10.1177/039463201402700303
- Stojkovska I, Wagner BM, Morrison BE. Parkinson’s disease and enhanced inflammatory response. Experimental Biology and Medicine. 2015;240(11):1387-1395. doi:https://doi.org/10.1177/1535370215576313
- Moon TC, Befus AD, Kulka M. Mast Cell Mediators: Their Differential Release and the Secretory Pathways Involved. Frontiers in Immunology. 2014;5. doi:https://doi.org/10.3389/fimmu.2014.00569
- Silver R, Curley JP. Mast cells on the mind: new insights and opportunities. Trends in Neurosciences. 2013;36(9):513-521. doi:https://doi.org/10.1016/j.tins.2013.06.001
- Theoharides TC, Alysandratos KD, Angelidou A, et al. Mast cells and inflammation. Biochimica et Biophysica Acta. 2012;1822(1):21-33. doi:https://doi.org/10.1016/j.bbadis.2010.12.014
- Ramesh G, MacLean AG, Philipp MT. Cytokines and Chemokines at the Crossroads of Neuroinflammation, Neurodegeneration, and Neuropathic Pain. Mediators of Inflammation. 2013;2013:1-20. doi:https://doi.org/10.1155/2013/480739
- Fernandez-Lizarbe S, Pascual M, Guerri C. Critical Role of TLR4 Response in the Activation of Microglia Induced by Ethanol. The Journal of Immunology. 2009;183(7):4733-4744. doi:https://doi.org/10.4049/jimmunol.0803590
- Amemori T, Jendelova P, Ruzicka J, Urdzikova L, Sykova E. Alzheimer’s Disease: Mechanism and Approach to Cell Therapy. International Journal of Molecular Sciences. 2015;16(11):26417-26451. doi:https://doi.org/10.3390/ijms161125961
- Levy D. Endogenous Mechanisms Underlying the Activation and Sensitization of Meningeal Nociceptors: The Role of Immuno-Vascular Interactions and Cortical Spreading Depression. Current Pain and Headache Reports. 2012;16(3):270-277. doi:https://doi.org/10.1007/s11916-012-0255-1
- Colton CA. Heterogeneity of Microglial Activation in the Innate Immune Response in the Brain. Journal of Neuroimmune Pharmacology. 2009;4(4):399-418. doi:https://doi.org/10.1007/s11481-009-9164-4
- Kunder CA, St John AL, Abraham SN. Mast cell modulation of the vascular and lymphatic endothelium. Blood. 2011;118(20):5383-5393. doi:https://doi.org/10.1182/blood-2011-07-358432
- Kawasaki T, Kawai T. Toll-Like Receptor Signaling Pathways. Frontiers in Immunology. 2014;5(461). doi:https://doi.org/10.3389/fimmu.2014.00461
- Gilfillan AM, Beaven MA. Regulation of Mast Cell Responses in Health and Disease. Critical ReviewsTM in Immunology. 2011;31(6):475-530. doi:https://doi.org/10.1615/critrevimmunol.v31.i6.30
- Shahid M, Tripathi T, Sobia F, Moin S, Siddiqui M, Khan RA. Histamine, Histamine Receptors, and their Role in Immunomodulation: An Updated Systematic Review. The Open Immunology Journal. 2009;2(1):9-41. doi:https://doi.org/10.2174/1874226200902010009
- Ferstl R. Histamine regulation of innate and adaptive immunity. Frontiers in Bioscience. 2012;17(1):40. doi:https://doi.org/10.2741/3914
- Kubo Y, Nakano K. Regulation of histamine synthesis in mouse CD4+ and CD8+ T lymphocytes. Inflammation Research. 1999;48(3):149-153. doi:https://doi.org/10.1007/s000110050438
- Parsons ME, Ganellin CR. Histamine and Its Receptors. British Journal of Pharmacology. 2009;147(S1):S127-S135. doi:https://doi.org/10.1038/sj.bjp.0706440
- Baronio D, Gonchoroski T, Castro K, Zanatta G, Gottfried C, Riesgo R. Histaminergic system in brain disorders: lessons from the translational approach and future perspectives. Annals of General Psychiatry. 2014;13(1). doi:https://doi.org/10.1186/s12991-014-0034-y
- Ennis M, Ciz M, Dib K, et al. Chapter 4 Histamine Receptors and Inflammatory Cells. Zurich Open Repository and Archive (University of Zurich). Published online October 22, 2013:103-144. doi:https://doi.org/10.2478/9788376560564.c4
- Liao TN, Hsieh KH. Characterization of histamine-releasing activity: Role of cytokines and IgE heterogeneity. Journal of Clinical Immunology. 1992;12(4):248-258. doi:https://doi.org/10.1007/bf00918148
- Lewis RA, Soter NA, Diamond PT, Austen KF, Oates JA, Roberts LJ. Prostaglandin D2 generation after activation of rat and human mast cells with anti-IgE. Journal of Immunology. 1982;129(4):1627-1631. doi:https://doi.org/10.4049/jimmunol.129.4.1627
- Haas HL, Sergeeva OA, Selbach O. Histamine in the Nervous System. Physiological Reviews. 2008;88(3):1183-1241. doi:https://doi.org/10.1152/physrev.00043.2007
- Arreola R, Becerril-Villanueva E, Cruz-Fuentes C, et al. Immunomodulatory Effects Mediated by Serotonin. Journal of Immunology Research. 2015;2015. doi:https://doi.org/10.1155/2015/354957
- Walker RF, Codd EE. Neuroimmunomodulatory interactions of norepinephrine and serotonin. Journal of Neuroimmunology. 1985;10(1):41-58. doi:https://doi.org/10.1016/0165-5728(85)90033-5
- Ahern GP. 5-HT and the immune system. Current Opinion in Pharmacology. 2011;11(1):29-33. doi:https://doi.org/10.1016/j.coph.2011.02.004